So, the first law is , where means the change in internal energy, is the heat, and is the work done. This is true whenever the thing we're paying attention to isn't losing or gaining particles, which we'll consider later. Let's check our understanding. Consider an air canister, like those used to blow dust out …
Category Archive: Thermo for Normals
Feb 14 2012
Thermo for Normals (part 9): Energy and temperature
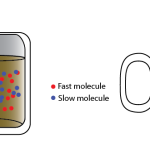
Energy and Temperature In the previous post I detailed the First Law of thermodynamics, which says that if something happens to a system or object, the thing called the internal energy of the object undergoes a change Don't pay attention to the actual equation. Just realize that the internal energy is increased by adding heat, …
Jan 24 2012
Thermo for normals (part 8): The first law of thermodynamics
When you are cold, one of the things you do instinctively is rub your hands together, or rub your arms with your hands. This warms them up by way of friction. That is, we have heated something up by motion. Caveman knew how to warm up his hands this way. He knew that rubbing made …
Jan 08 2012
Thermo for normals (parts 6-7): Evaporation, boiling, and condensation
Thermo for Normals is a short book (~100 pages) I wrote that conveys the basics of thermodynamics without all of the mathematical complexity. Over the next year I will be posting 1 or 2 sections of the text per week. I don't know whether I will ever try to publish it in any other form, …
Dec 19 2011
Thermo For Normals, Part 5 (of 30)
Thermo for Normals is a short book (~100 pages) I wrote that conveys the basics of thermodynamics without all of the mathematical complexity. Over the next year I will be posting 1 or 2 sections of the text per week. I don't know whether I will ever try to publish it in any other form, …