So, the first law is 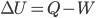 , where
, where 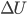 means the change in internal energy,
means the change in internal energy,  is the heat, and
is the heat, and 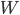 is the work done. This is true whenever the thing we're paying attention to isn't losing or gaining particles, which we'll consider later.
is the work done. This is true whenever the thing we're paying attention to isn't losing or gaining particles, which we'll consider later.
Let's check our understanding. Consider an air canister, like those used to blow dust out of computers. The air that comes out of it is cold. Why? The First Law says that 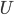 will go down (and hence temperature will go down) if either
will go down (and hence temperature will go down) if either  goes out or the system does work
goes out or the system does work 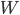 on something. Is there heat involved in letting the air out of the canister. No. Is the air doing work as it leaves? Yes! It has to push the air in front of it out of the way as it expands. That lowers its internal energy and its temperature. If you discharged that gas into a vacuum, the temperature would not go down.
on something. Is there heat involved in letting the air out of the canister. No. Is the air doing work as it leaves? Yes! It has to push the air in front of it out of the way as it expands. That lowers its internal energy and its temperature. If you discharged that gas into a vacuum, the temperature would not go down.
But what about if you want to just use electricity and cycle something to make cold, such as your refrigerator and air conditioners do? There is no such thing as "cold". There's heat, which is the transfer of energy into something's internal energy. But there is no anti-heat. Burning something makes heat, but there's no simple way to make "cold". It's hard to see how you can build a closed-cycle refrigerator, where no atoms leave. (Try to think of how you would do that. I'll wait.) Here's an idea: 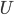 will decrease (
will decrease (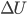 will be negative) if we could find a way to let the system do some work without heating it (
will be negative) if we could find a way to let the system do some work without heating it ( but
but 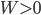 ). And if
). And if  decreases, then temperature does too. That's the same as the air expanding from the air can. But we can also do this by use of a piston.
decreases, then temperature does too. That's the same as the air expanding from the air can. But we can also do this by use of a piston.
Here is the schematic way that almost all refrigerators and air conditioners work:
- Start with some substance (gas) in a vessel that can control its volume (a piston).
- Compress the gas. In doing so you will heat it up because you did work on it. (This is why the outside unit in your AC is called a compressor.)
- Now let it come to equilibrium with the outside air by heat conduction. Heat
 will leave the gas, reducing its internal energy, and making its temperature roughly the same as the outside air's (this is why AC compressors have a big fan in them. It's also why the AC compressor has to be located outside: you don't want to dump that heat into your house. Otherwise you'll actually heat your house instead of cooling it!)
will leave the gas, reducing its internal energy, and making its temperature roughly the same as the outside air's (this is why AC compressors have a big fan in them. It's also why the AC compressor has to be located outside: you don't want to dump that heat into your house. Otherwise you'll actually heat your house instead of cooling it!) - Since the pressure is now higher inside the piston than outside, let the piston expand. The gas will do some work, lowering its internal energy and hence its temperature. You have now made the gas in the piston cold. Place whatever it is you'd like to be cold (like a soda, or your house) in contact with the piston, which will absorb heat as long as its temperature is lower than what you want to cool down.

But is making our soda cold all that we've done? We made the soda cold, but what about the whole planet, or the universe? This I leave to another day, but note that we had to throw some heat  into the atmosphere, and moreover somebody had to do some work from the outside to compress the gas in the first place.
into the atmosphere, and moreover somebody had to do some work from the outside to compress the gas in the first place.
Now, going back to the first example, we can make something cool by losing particles. Another type of in-home cooling is an evaporative cooler or a "swamp cooler". This is an extremely simple device. You have a mesh that is coated in water, and you blow air through the mesh. The air that comes through is cooler than went in, though it is slightly moister. Why the heck does this work?
The first thing to realize is that the overall speed of the air is tiny compared to the speed of the molecules in it. It's sort of like carrying a box of gas to another location, in contact with water, and the carrying it away again. Some of the water evaporates into the air as it passes through. The vapor is at a relatively low temperature, since the molecules gave up their energy in the jumping out. The air is now mixed with a cooler water vapor, reducing the temperature.
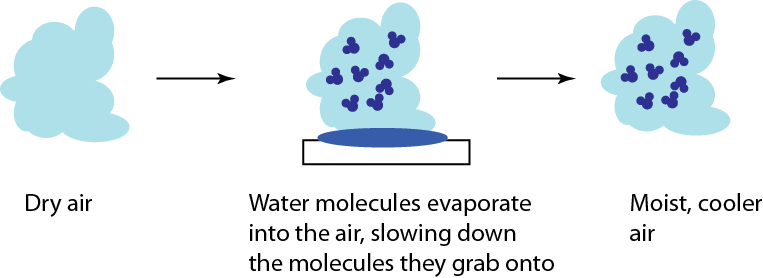
An evaporative cooler won't work well if the air is already saturated with water. Lots of water in the air makes evaporation harder, since for every molecule that evaporates, another one that was already vapor condenses on the water. So you would never use a swamp cooler in a swamp! Dry environments work best. (Conventional wisdom is that the name "swamp cooler" comes from the fact that these machines tend to gather mildew, making them smell like a swamp.)





