Energy and Temperature
In the previous post I detailed the First Law of thermodynamics, which says that if something happens to a system or object, the thing called the internal energy of the object undergoes a change

Don't pay attention to the actual equation. Just realize that the internal energy is
- increased by adding heat,
- decreased if the system does work, and
- increased if particles are added
And if the internal energy per particle (the average energy) increases, so does the temperature.
Modern folk have a big advantage over people 150 years ago when thinking about thermodynamics, in that we know that all matter on Earth is made up of atoms. The internal energy is therefore understandable from an elementary point of view: the atoms are whizzing around (in a gas), jiggling vigorously (in a liquid), or jiggling a bit (in a solid).
Now I want to talk about what temperature is. An object or system is made up of a bunch of atoms. Let's discuss a cup of hot coffee as our example. It's a liquid, and so the particles are all moving around, and they knock into each other a lot. Each particle has some speed, and hence a kinetic energy 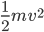 . All of their energies are different, but we don't really care what a given atom's energy is; we only care about what the average is. (Later we might be interested in more details.)
. All of their energies are different, but we don't really care what a given atom's energy is; we only care about what the average is. (Later we might be interested in more details.)
Think of the atoms in the liquid as people in the United States, and the amount of kinetic energy the atoms have as the amount of money the people have. Then, the temperature is determined by how rich the average citizen is. In physical terms, take the total energy of all the atoms and divide it by the number of particles---the higher that average is, the higher the temperature.
Let's see, from some examples, whether or not this analogy holds.
Suppose that one day Canada was annexed by the US. Since Canada is roughly as wealthy as we are, the average wealth would be about the same in the newly expanded US as it was before it added the new territory. The total amount of money increased, but the number of people increased proportionally.
Similarly, if you combine two cups of coffee, both at the same temperature, the resulting mixture has the same temperature as the two cups had before. The total internal energy 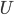 went up, but the number of particles went up proportionally, so the average energy stayed the same. Thus, there is no change in temperature. This is, of course, what we expect if we add liquid at the same temperature.
went up, but the number of particles went up proportionally, so the average energy stayed the same. Thus, there is no change in temperature. This is, of course, what we expect if we add liquid at the same temperature.
Now, instead, suppose the US annexed Mexico. The total wealth would increase, but the number of people coming in is greater per dollar. You've added one group with a high average wealth to another with a (much) lower wealth. As a consequence, the average wealth decreases.
So, if we add cool cream to our coffee, what happens? Of course, the mixture is cooler! But by that, we specifically mean that the temperature decreased. We added a substance with relatively low average energy to one with high average energy, which drags the overall average, and hence temperature, down.
But your coffee can also cool down when it's just sitting on your desk. How does that work?
When we sit down for a cup of coffee (usually very hot in order to brew properly) we have to wait for it to cool. You might quickly realize that there is steam coming off of it, or you might notice that the air above the coffee is much warmer than room temperature and it's "clammy", which is to say that if you hold your hand over it for awhile and take it away your hand will be damp. What you feel is not only hot air, but actual water molecules in vapor form. But why exactly does this cool the liquid down?
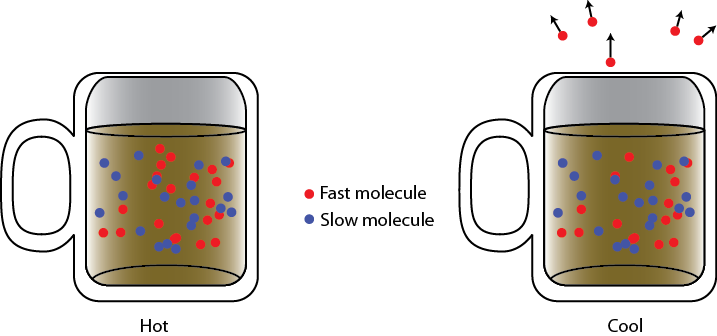
What is happening is that some of the atoms in the coffee have a higher energy than other ones. The atoms in the coffee with the highest energy jump out of the water, forming a hot layer of air and water vapor just above the cup. Just removing atoms doesn't of itself reduce the temperature. However, since the fastest atoms are the ones that jump out, the internal energy per particle (which is to say, the average energy of the particles) decreases, and this does reduce the temperature. If you want to speed the cooling up, you should remove the layer of hot air/vapor above the cup as soon as it develops. If you do this, then you make it possible for the next-fastest molecules to escape, reducing the temperature yet more. Removing the lid helps a lot, as it gives a lot more molecules the change to drift away from the cup, making room for new water molecules to take their place. Blowing over the top of the cup helps too, as you quickly get the hot atoms out of the way. So this is how coffee cools. Of course, you knew that! Blowing on stuff cools it. But the reason that it works is that the fast atoms jump out, you clear them away immediately by blowing, and then the next fastest jump out, etc. Each time you do this, the temperature drops because the average energy drops.





