When you are cold, one of the things you do instinctively is rub your hands together, or rub your arms with your hands. This warms them up by way of friction. That is, we have heated something up by motion. Caveman knew how to warm up his hands this way. He knew that rubbing made things warmer. He knew that if he struck a flint or rapidly turned a stick against tinder he could make fire. In short, caveman knew a simple form of the First Law of Thermodynamics:
In light of this, then, you may wonder why scientists until the mid-19th century believed that heat was a thing, little "particles of warmth" called caloric. They thought when you burned wood you were releasing its caloric, and the pot of water you were boiling was absorbing the caloric. The more caloric you had, the higher the temperature would go. The theory, though wrong, had an internal consistency when it came to analyzing the operation of steam engines, the popular topic of the day. And it was certainly appealing: we can all get our heads around little particles flowing from one place to another.
At the same time, people were developing the idea of energy. Energy, to them, was nothing other than the ability to push things around and get them moving. Two kinds of energy were recognized: kinetic energy (the energy associated with a thing moving) and potential energy (stored energy against a force field like gravity). If you were moving, you could crash into something and make it move (do work on it), and if you had already done work lifting a weight against gravity, that work you had done would translate into the motion of the weight when you let it go. There was no real identification of other forms of energy, such as chemical, thermal, or electromagnetic, that we now talk about.
As we know, energy is something that is conserved. If you have some energy in a box, and you don't let any energy into it or let energy out of it, then the stuff in the box has some amount of energy in it (say, for instance, 50 joules), and that amount never changes. We also say that mass is conserved, so that if you have some oxygen molecules in a room, no matter what you do chemically, the oxygen atoms are always somewhere. Maybe they each joined with 2 hydrogens to make water, or were consumed while something burned to make carbon dioxide, or whatever; but if you look carefully, you will find the atoms somewhere: oxygen atoms are conserved.
Well, at the time, people had no real expectation that energy was conserved! The very first physics problem you can think of demonstrated no apparent properties of energy conservation. Suppose I hit a baseball into the air. Where did the energy that made the motion of that baseball come from? Well, it came from me, but where did I get it? I wasn't moving, I didn't have any identifiable potential energy. It seems that I created the energy out of nothing. Now the ball lifts into the air and momentarily stops at the apex of its trajectory. All the kinetic energy has been translated into potential energy. This was understood. But once the ball falls back to earth, it strikes the ground and stops. Where did the energy go? It seems that it was destroyed when the ball hit the ground. The box is the baseball stadium, and some energy inside it was created and then destroyed. Energy, it seemed, wasn't conserved.
When people thought about thermodynamics in the 19th century, they thought about steam engines. Trains, riverboats, these were objects of thermodynamics. And both had one thing in common: they involved the boiling of water. And everybody knows how you boil water. You burn something like coal, wood, or oil underneath a pot of water, and after awhile, the water heats up and boils. The more heat you added to the water, the more steam came off. Energy? That didn't seem to come into play.

Benjamin Thompson (Count Rumford), 1753-1814
In light of this, why not believe in something called caloric? Isn't it reasonable that the things you use to heat (oil, wood, coal) have caloric in them, and when you burn them the caloric goes into the water?
But one day a guy, Benjamin Thompson, was engaged by the British government to make cannons for its army. Because cannons needed to be very smooth on the inside, you couldn't just cast them in iron. You cast the general shape, and then used a very big drill to bore out the firing chamber. That way cannonballs would run down the muzzle in a smooth way and not blow the cannon apart.
Anybody who's ever worked with metal knows that you have to keep it cool while cutting it, so Thompson kept water in the cavity as he drilled it out. While boring out the cannon, he put a thermometer inside it, curious as to what happened to the liquid during the process. The story goes that to his surprise, the water's temperature was increasing. After 2 1/2 hours of drilling, the water actually came to a boil.
Though Thompson soon lost interest in the problem, James Prescott Joule knew that there was something to it, and endeavored to be more precise. What he thought was that if energy was put into a system in some way, and that energy was no longer visible at a later time, that the energy must show up as heat. This is simple to prove: He devised the machine depicted in this video:
Two weights were lifted into the air by the operator, giving the system a potential energy  . When these weights dropped, they turned a paddle inside a container of water. In each cycle, Joule observe that the temperature increased by the same amount! This was indisputable proof of the following statement of the First Law:
. When these weights dropped, they turned a paddle inside a container of water. In each cycle, Joule observe that the temperature increased by the same amount! This was indisputable proof of the following statement of the First Law:
If a system appears to lose energy, in reality the energy went into heating the object.
This changes everything. Going back to the previous example of hitting a baseball, the energy "created" is actually converted from the chemicals in the player's body by being burned. The energy "destroyed" actually shows up as a rise in the temperature of the baseball and the earth where the ball struck the ground. The whole system had some amount of energy, and it was the same throughout. Energy is conserved.
It was also realized that temperature, which everyone has some notion of, corresponds somehow to how much energy is inside a thing. We could call it the internal energy of the object. Take for instance the baseball. It has a certain temperature, which is about room temperature. What this implies is that it has an internal energy that corresponds to the temperature. If you tell me the temperature, I can tell you how much energy is stuffed into it. This has nothing to do with it moving (which would be kinetic energy  ) or potential energy (
) or potential energy ( ) which are about the motion of the whole object. This was energy the object had stuffed inside it when it's just sitting on the ground doing nothing---the vibrations of particles that it's made of.
) which are about the motion of the whole object. This was energy the object had stuffed inside it when it's just sitting on the ground doing nothing---the vibrations of particles that it's made of.
From this point of view, denial of atomic theory (that all matter is made up of atoms) began to seem preposterous. For surely somehow this internal energy was in fact the kinetic and potential energy of the little bits inside the object.
With heat, work, and internal energy, we can enunciate the first law in a semi-technical form:
If no particles leave}, then 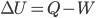 , where
, where 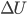 is the change in internal energy,
is the change in internal energy,  is the heat added, and
is the heat added, and 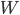 is the work done by the system.
is the work done by the system.







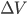
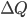
1 comment
rosaria
1/24/2012 at 2:46 PM (UTC -4) Link to this comment
i didn't know you had a blog!
Thanks for sharing.